Weathering, Erosion, Deposition Powerpoint:
Prepare for your test using this ppt and notes from class-
1st 6 Weeks Key Vocabulary for 5th Grade Science
Ms. Starling's CHALLENGE: Make up a fiction Science Story that involves at least 10 of these definitions! Illustrate your story!
Key Content Vocabulary:
- Boiling point – the temperature at which a substance changes from a liquid to a gas; for water this temperature is 100°C
- Celsius – the international scale of measurement for temperature
- Conductor – a substance or object that allows energy to flow through it easily
- Dissolving – the process by which substances breakdown into small pieces and spread evenly in liquid (water)
- Electrical energy – energy that is absorbed or delivered by an electric circuit
- Insulator – a substance or object that does not allow energy to flow through it easily
- Magnetism – a force of attraction that causes a magnetic material to move
- Mass – the amount of matter in something
- Matter – anything that has mass and takes up space
- Melting / freezing point – the temperature at which a substance changes from a liquid to a solid or a solid to a liquid by the loss or addition of heat; for water this temperature is 0°C
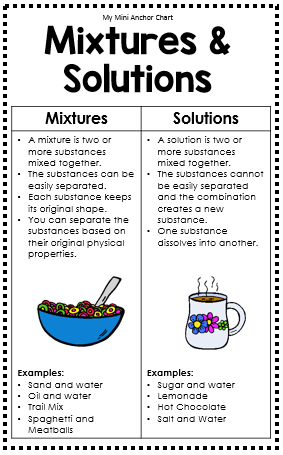
- Mixture – a combination of two or more substances that can be easily separated in some physical way
- Physical properties – properties of matter that can be observed, measured, or changed without changing the matter itself
- Properties – characteristics used to identify matter
- Relative density – floating or sinking when compared to water
- Solubility – the ability of a substance to dissolve in another substance, such as sugar dissolving in water
- Solution – a type of mixture in which the particles of one or more substances are uniformly dispersed (spread out) throughout another substance
- Temperature – a way of measuring how hot or cold something is; temperature is measured using either the Fahrenheit (F) or Celsius (C) scale
- Thermal / heat energy – energy related to the temperature of an object or a substance
- Volume – the amount of space that a substance or object takes up.
Density
https://www.youtube.com/watch?v=SimFy9wOMXY
Density Youtube Video
- Explore density at home: Fill up a cup with water. Does a raw egg float or sink? Add salt to the water until the egg floats. How does this affect density? Record your findings!
- Make another density tower! Show your parents, siblings, or friends!
Density Youtube Video
- Explore density at home: Fill up a cup with water. Does a raw egg float or sink? Add salt to the water until the egg floats. How does this affect density? Record your findings!
- Make another density tower! Show your parents, siblings, or friends!
Mixtures and Solutions
Check out the Quizlet flashcards and tasks for Mixtures and solutions:
https://quizlet.com/15601269/mixtures-and-solutions-stemscopes-flash-cards/
BILL NYE!!!!!!!!!!! Mixtures and Solutions
Mixtures and Solutions Study Guide
1 What is a mixture?
a. A combination of two or more substances that does not produce a new substance
2 What is a solution?
a. A mixture with one substance spread out so evenly in another substance that you cannot tell the two substances apart
3 Is a solution a mixture or is it just a solution?
a. A solution is a type of homogenous mixture
4 What is easier to separate a mixture or a solution?
a. Mixture
5 What are the physical properties and how to they relate to mixtures?
a. Properties that can be observed, measured, or changed without changing the substance itself
6 List 5 examples of mixtures
a. Chili powder and iron fillings
b. Marbles and paper clips
c. Chex Mix
d. Aluminum cans and iron cans
e. M&Ms and Buttons
7 List 5 examples of solutions
a. Koolaid
b. Lemonade
c. Salt and Water
d. Soda
e. Coffee
8 What does dissolve mean?
a. the process by which substances breakdown into small pieces and spread evenly in liquid (water)
9 What is a solute? Give an example
a. The item being dissolved. Ex: Salt, sugar
10 What is a solvent? Give an example
a. The liquid; what the solute is dissolved in. Ex: water
11 What is the universal Solvent?
a. Water
12 What does a saturated solution look like?
a. Containing the largest possible amount of solute
(when a liquid cannot dissolve anymore of a substance)
13 What is the best way to separate a solution?
a. Filtration
14 What tool/process would help you separate the following:
- iron nails from sand?
o Magent
- What about cork pieces from sand?
o Sieve
- What about salt from water?
o Filter
- What about sand and water?
o Filter
- Marbles and toothpicks?
o Water
15 How does salt react with water?
a. Salt dissolves in water
16 How does pepper react with water?
a. Pepper does not dissolve in water
17 How do you dilute a solution?
a. Add solvent (liquid)
18 Draw a picture of citric acid crystals. Draw a picture of salt cyrstals.
a. Crystals: swirls, line
b. Salt: tiny squares
1 What is a mixture?
a. A combination of two or more substances that does not produce a new substance
2 What is a solution?
a. A mixture with one substance spread out so evenly in another substance that you cannot tell the two substances apart
3 Is a solution a mixture or is it just a solution?
a. A solution is a type of homogenous mixture
4 What is easier to separate a mixture or a solution?
a. Mixture
5 What are the physical properties and how to they relate to mixtures?
a. Properties that can be observed, measured, or changed without changing the substance itself
6 List 5 examples of mixtures
a. Chili powder and iron fillings
b. Marbles and paper clips
c. Chex Mix
d. Aluminum cans and iron cans
e. M&Ms and Buttons
7 List 5 examples of solutions
a. Koolaid
b. Lemonade
c. Salt and Water
d. Soda
e. Coffee
8 What does dissolve mean?
a. the process by which substances breakdown into small pieces and spread evenly in liquid (water)
9 What is a solute? Give an example
a. The item being dissolved. Ex: Salt, sugar
10 What is a solvent? Give an example
a. The liquid; what the solute is dissolved in. Ex: water
11 What is the universal Solvent?
a. Water
12 What does a saturated solution look like?
a. Containing the largest possible amount of solute
(when a liquid cannot dissolve anymore of a substance)
13 What is the best way to separate a solution?
a. Filtration
14 What tool/process would help you separate the following:
- iron nails from sand?
o Magent
- What about cork pieces from sand?
o Sieve
- What about salt from water?
o Filter
- What about sand and water?
o Filter
- Marbles and toothpicks?
o Water
15 How does salt react with water?
a. Salt dissolves in water
16 How does pepper react with water?
a. Pepper does not dissolve in water
17 How do you dilute a solution?
a. Add solvent (liquid)
18 Draw a picture of citric acid crystals. Draw a picture of salt cyrstals.
a. Crystals: swirls, line
b. Salt: tiny squares